 
And we should find that that is equal to 5.659 times 10 to the negative, 23 cubic centimeters. The values given above are inserted into the equation below: Dm 65 / 10 6.5 (kg/m3) Example Problem 2: For this problem, the variables needed are provided below: metal mass (kg) 77. Finally, calculate the Metal Density using the equation above: Dm MM / mV. Relative atomic massThe mass of an atom relative to that of carbon-12. The metal volume (m3) is provided as: 10. Um, given in the question, um, and knowing the mass of the radio immune itself, we can rearrange this equation in order to determine the volume of the already immune itself. Density (g cm3)Density is the mass of a substance that would fill 1 cm3 at room temperature. Calculate the density of an iridium sample. The atomic radius of the iridium atom is (1.33e - 8) cm. Iridium metal crystalizes in a body centered cubic unit cell. I have developed an Excel® based software tool to perform these calculations. Hence the weight fraction of copper is 1-0.958 0.042. Substituting these values into equation 2 gives the weight fraction of gold as 0.958. Calculate the density of an iridium sample. The density of gold (metal A) is 19.32 g/cc and that of copper (metal B) is 8.92 g/cc. The atomic radius of an iridium atom is 1.33e-8 cm. 
So density is equal to mass over volume in knowing the density of the already immune itself. Iridium metal crystalizes in a body-centered cubic unit cell. And we can do that by multiplying the molar mass of iridium, which we know from the periodic table, um, dividing that by avocados number in order to get the mass of, um, just one iridium Adam and then subsequently multiplying that by the total number of atoms in an Iridium unit cell, which is for and we should find that it is equal to 1.277 times 10 to the negative 21 g. Iridium has an atomic weight of 192.22 g/mol and an atomic density of 22.56 g/cm³. And this means that in an Iridium unit cell, there are a total of four atoms, so we can begin to calculate the radius of the radium Adam by first calculating the mass of the radium Yoon itself. Use Avogadros number to calculate the atomic mass of iridium. 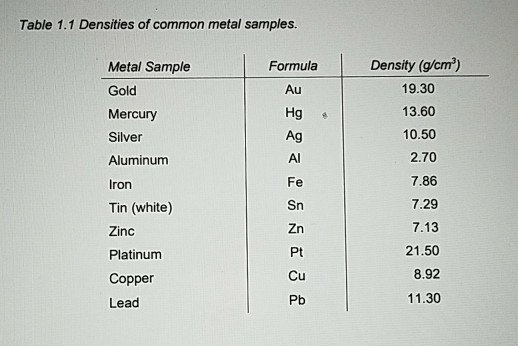
The edge length of the unit cell was found by x-ray diffraction to be 383.9 pm. So this question wants us to determine the radius oven or idiom. Iridium metal, Ir, crystallizes in a face-centered cubic (close-packed) structure. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
